
-
Lean NO_x Trap (LNT) catalyst
Lean NO_x Trap (LNT),also known as NOx Adsorber Catalyst (NAC), is a clever "storage-and-release" system used primarily in diesel engines to clean up Nitrogen Oxides (NOx) without needing an external fluid like urea (AdBlue).
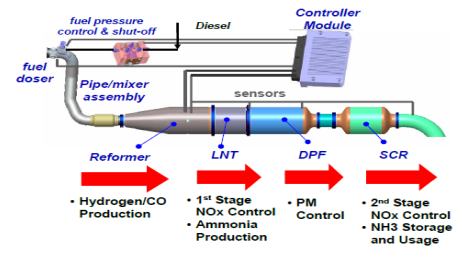
Combination system between LNT, SCR and DPF
If SCR is like a continuous chemical spray, LNT is more like a chemical sponge that soaks up pollutants and then squeezes them out when it's time to clean them.
1. How It Works:
The LNT operates in a constant cycle between two distinct engine states. The 1st one is the Storage stage(Lean Operation).
During normal driving, diesel engines run "lean" (lots of air, little fuel).
Oxidation: The Nitric Oxide (NO) in the exhaust is oxidized into Nitrogen Dioxide (NO2) by a precious metal catalyst PGM(usually Pt/Pd).
Adsorption: This NO2 reacts with an alkaline earth metal (usually Barium Oxide, BaO) on the surface of the catalyst to form Barium Nitrate [Ba(NO3)2].
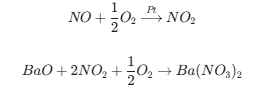
Result: The NOx is "trapped" on the catalyst surface instead of going out the tailpipe. This usually lasts for 30 to 60 seconds.
Phase 2: The Regeneration Phase (Rich Operation)
Once the "sponge" is full, the engine’s computer briefly changes the combustion to rich condition (excess fuel, very little oxygen) for just 2 to 5 seconds.
Release: The excess fuel creates a "reducing" environment (CO, H2, and HC). This makes the Barium Nitrate unstable, releasing the NOx.
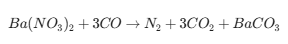
Reduction: The released NOx immediately reacts with the CO and H2 over a Rhodium (Rh) catalyst.
Result: The NOx is converted into harmless Nitrogen (N2) and water vapor.
2. The Catalyst Composition
To make this happen, the LNT is coated with a complex "washcoat" consisting of:
l Oxidation Catalyst: Platinum (Pt) to turn NO into NO2.
l Adsorbent: Barium (Ba) to store the NOx.
l Reduction Catalyst: Rhodium (Rh) to finish the job and produce N2.
l Support: Alumina (Al2O3) to hold everything in place.
3. Sulfur Poisoning and Thermal Durability
LNTs rely on rear earth metals, primarily Barium Oxide (BaO), to store NOx as nitrates. The fundamental weakness is that Barium has a much higher chemical affinity for Sulfur (S) than for Nitrogen (N).Sulfur species (SOx) from fuel and lube oil react with the storage sites to form Barium Sulfate (BaSO4).like nitrates, which decompose at 350℃, sulfates are thermally stable and will not release the storage site during normal engine operation. This leads to a linear decline in NOx Storage Capacity (NSC), eventually rendering the catalyst useless.
As the catalyst ages or sulfur levels increase, DeSOx cycles must occur more frequently. Therefore,more frequent DeSOx cycles accelerate thermal aging, which in turn reduces the catalyst's tolerance to sulfur, leading to a rapid end-of-life. Meanwhile, the rich-burn spikes required for regeneration and the high-heat cycles for DeSOx impose a significant Fuel Economy Penalty and increase the thermal stress on the engine exhaust manifold.
4. DOC vs LNT
The DOC is the most basic part of a modern diesel exhaust system.It simply "burns" (oxidizes) the invisible gases. A DOC does not reduce NOx to N2. In fact, it converts NO into NO2. However, this is actually helpful for the DPF (Diesel Particulate Filter) downstream, but it doesn't lower the total NOx count.
On the other hand, the LNT is essentially a DOC on steroids. It contains the same precious metals (Pt/Pd) as a DOC, but it adds a storage element(Ba) to trap the NOx in the low temperature condition.
To sum up, a DOC is a "continuous incinerator," while an LNT is a "smart sponge" with an incinerator built-in.
The primary job of a DOC is to "burn off" hydrocarbons (the diesel smell) and toxic Carbon Monoxide (CO). However, it has zero capacity to remove Nitrogen Oxides (NOx)—the specific pollutant that Euro 6 regulations target most strictly. In contrast, an LNT is a "Smart Sponge" specifically engineered to catch and eliminate NOx.
An LNT is a much more complex device than a DOC due to its specialized chemical coating:
The Sponge (Barium): Unlike a DOC, the LNT contains a thick layer of Barium salts. This acts like a magnet, trapping NOx molecules during normal driving so they don’t exit the tailpipe.
The Cleaner (Rhodium): LNTs contain high levels of Rhodium. When the "sponge" is full, the engine performs a "rich-burn" cycle to wash the catalyst. The Rhodium then converts the trapped toxic gas into harmless Nitrogen (N2).
5. Why You Cannot Replace an LNT with a DOC on Euro 6 Vehicles?
Although a DOC (Diesel Oxidation Catalyst) and an LNT (Lean NOx Trap) may look identical on the outside, they perform completely different tasks inside your exhaust system.
Modern Euro 6 vehicles are equipped with highly intelligent OBD and downstream NOx sensors.If you install a DOC, the sensors will immediately detect that NOx levels are over the legal limit, triggering the Emission Fault Light on the dashboard.
The computer will realize it cannot perform the periodic "regeneration" (cleaning the sponge). To protect the environment, the vehicle may enter Limp Mode, severely limiting engine power and causing fuel consumption to skyrocket.
Expert Advice:
Navigating Euro VI emission standards requires precision and the right hardware. If you have any questions regarding Euro 6 catalysts or emission compliance, please consult the experts at TES. We are here to ensure your fleet stays efficient and compliant.